PAD
Dedicated to testing and early intervention
Peripheral Artery Disease (PAD) is a common, yet serious, disease that is often not diagnosed but can be treated.
Learn More
Women's Health
Non-surgical treatments in a caring environment by people who listen
We offer a minimally invasive treatment options for various women's health conditions including uterine fibroids, bleeding internal hemorrhoids, pelvic venous insufficiency/congestion, osteoarthritic joint pain, among other.
Learn More

Men's Health
Non-surgical treatments to help you feel better and improve your quality of life
We offer a minimally invasive treatment options for various men's health conditions including benign prostatic hypertrophy, bleeding internal hemorrhoids, varicocele, and osteoarthritic joint pain, among others.
Learn More
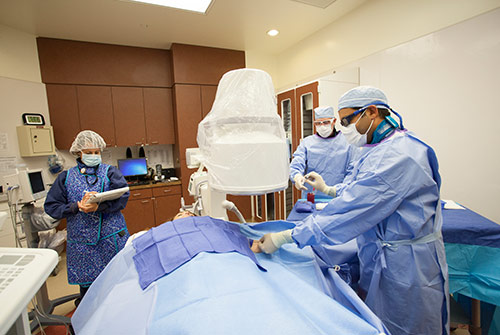
Our Services
Minimally Invasive Treatment Options for Vascular Conditions
Our outpatient vascular access center in Oakbrook, IL offers a complete range of minimally invasive vascular procedures, including treatment options for Peripheral Artery Disease (PAD), Uterine Fibroids and Dialysis Access Management.

Our Doctors
Meet our team of highly trained professionals
Our physicians are skilled, experienced specialists in vascular and interventional radiology. In addition to offering the highest quality medical care and latest techniques, we care about the comfort and well-being of our patients, treating them like family.
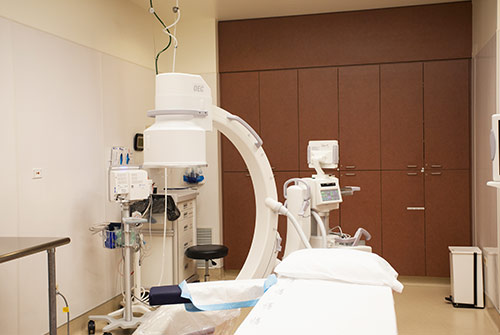
Our Facility
State-of-the-art Outpatient Vascular Center
All procedures are performed in our state-of-the-art outpatient interventional and vascular radiology center, conveniently located in Westmont, IL. Our facility is accredited by The Joint Commission.








